Unknown chemicals in the environment
Chemicals like pesticides and plastics provide us with a comfortable and convenient daily life. The price of spinach might go up without the use of pesticides. Game consoles and smart phones might not exist without plastics. On the other hand, some of these chemicals have toxic effects and their usage should be carefully managed. Hence, in order to minimize their risks, various safety tests are mandated prior to their industrial production and import. One such test is a degradability test, mandated to see the degree of degradation of a chemical.
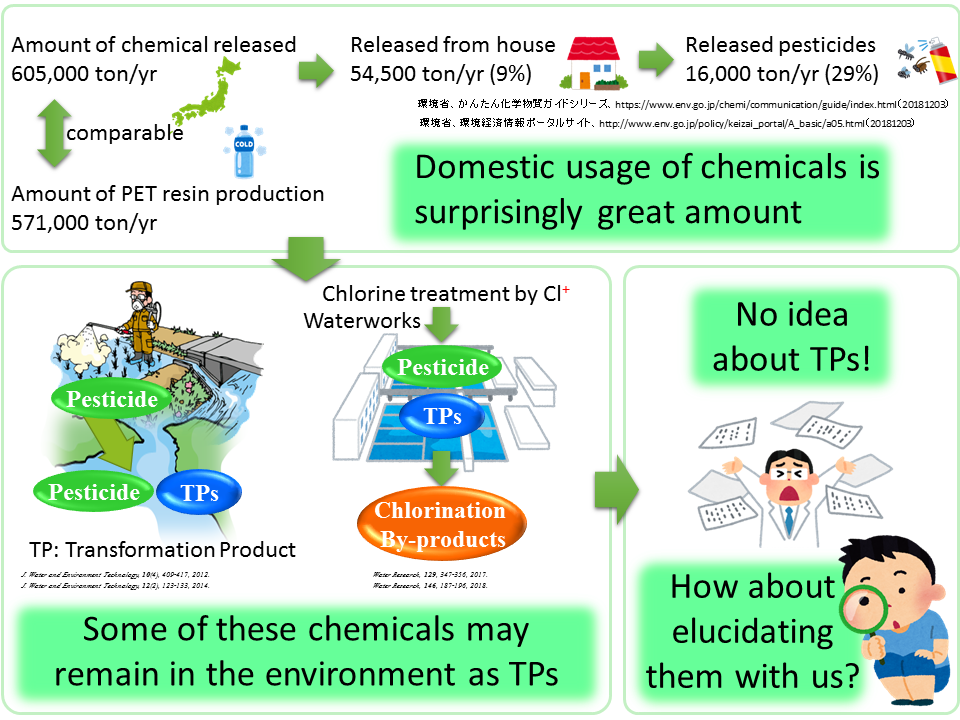
As shown in the figure below, the amount of chemicals released into the environment in FY2005 was estimated to be 605,000 ton/yr in Japan, according to Ministry of the Environment, Japan. Of this, 54,500 ton/yr are released from houses and the amount of pesticides released accounted for the 29% of the value (16,000 ton/yr). Don’t you think that’s a surprisingly large amount?
Chemicals released into the environment generally degrade and are eventually converted into simple substances, e.g. carbon dioxide or water, however, some of them undergo incomplete degradation and their transformation products (TPs) such as hydrolysates and oxidates will be generated. Chlorination of these TPs at waterworks can lead to their further conversion into chlorination by-products. It is thus necessary to elucidate the chemical structures for the assessment of their environmental risks, but often, little or no information is available on these chemicals.
How would you like to elucidate chemicals with us?
How to elucidate unknowns
Structural elucidation of an emerging contaminant is generally a formidable challenge because of its low concentration in the environment. To make matters worse, coexisting natural organic matter may disturb the elucidation. Liquid chromatograph-high resolution mass spectrometer (LC-HRMS) is one of the most promising way to overcome this problem. Thus we have elucidated some unknowns with LC-HRMSs.
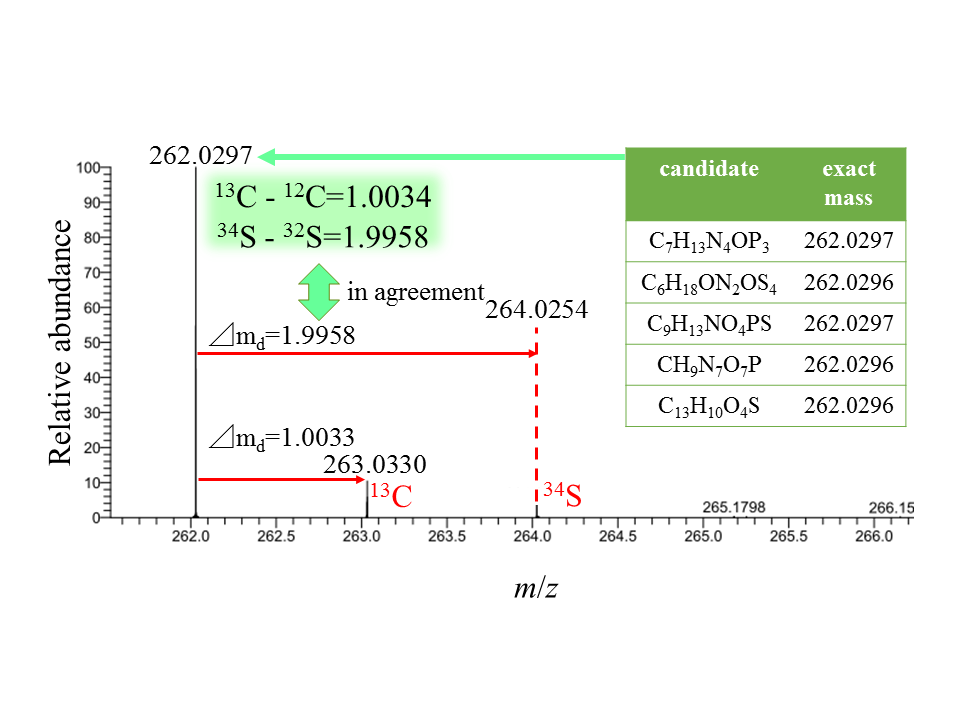
You may have learned the atomic weight of the oxygen atom as 16, but its exact mass is 15.99491461957. Our technique can determine the exact mass of oxygen as 15.9949. Exact masses of other atoms are also determined with the same level of precision. Such high precision can narrow down the number of possible molecular formulas of an ion and can give us an unequivocal formula. For instance, if the exact molecular weight (literally, m/z) of an unknown compound is 262.0297, its ion formula may be C7H13N4OP3, C6H18ON2OS4, C9H13NO4PS, CH9N7O7P or C13H10O4S. Factoring in stable isotopes as well as the m/z value, an unequivocal formula can be derived. In this case, the relative abundances of 13C and 34S indicate that the number of carbon and sulfur in the ion should be nine and one. The ion formula of the unknown therefore can be determined as C9H13NO4PS. LC-HRMS is a powerful tool for elucidating an unknown.


